
EUDAMED: A database to strengthen European market surveillance and increase transparency
EUDAMED stands for European Database on Medical Devices, and is a web-based portal acting as a central hub for the exchange of information between national competent authorities of the EU Member States and the European Commission [12]. EUDAMED is an integral part of implementing the two Medical Devices Regulations.
Based on the European Commission decision of 19. April 2010 [1], the European Databank on Medical Devices (EUDAMED) was established as a database for the purposes of Article 10b(3) of Directive 90/385/EEC, Article 14a(3) of Directive 93/42/EEC and Article 12(3) of Directive 98/79/EC. According to Article 6, Member States should have applied this decision from 1. May 2011 and data should have been entered by 30. April 2012 at the latest.
![Figure 1: Header of the EUDAMED Website [2]](https://biomatdb.eu/wp-content/uploads/2022/12/BiomatDB-EUDAMED-A-database-to-strengthen-European-market-surveillance-and-increase-transparency-img-01.jpg)
The purpose of the EUDAMED electronic database is stated in Article 33 Medical Device Regulation (MDR), which requires that the Commission takes responsibility for establishing the system that should “…enable the public to be adequately informed about devices placed on the market…” [3]. EUDAMED was thus created to strengthen market surveillance and transparency regarding all medical devices placed on the European market. Its creation is one of the key aspects of the MDR (EU) 2017/745 [4] and in vitro diagnostic MDR (EU) 2017/746 [5].
According to the home page, “EUDAMED will provide a living picture of the lifecycle of medical devices that are made available in the European Union (EU). It will integrate different electronic systems to collate and process information about medical devices and related companies (e.g., manufacturers). In doing so, EUDAMED aims to enhance overall transparency, including through better access to information for the public and healthcare professionals, and to enhance coordination between the different Member States in the EU” [2].
User experiences
A survey [6] amongst the Member States, published in October 2012, shed light on how the main users experienced EUDAMED. Some interesting findings are shown below.
There were indications of under-reporting in actors (with eleven Member States having recorded less than 100) and numbers of devices (with ten Member States having recorded less than 500).
Although data entered after May 2011 was found to be accurate by 92%, 6% of mandatory fields were filled in with ‘NA’ while, on average, 42% of the optional fields were used (as shown in figure 2 below).
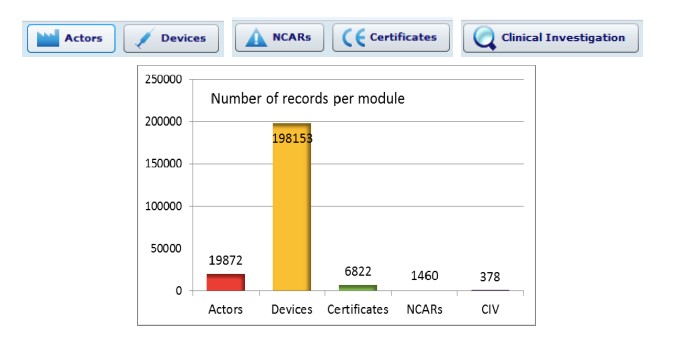
![Figure 2: Evaluation of the “European Databank on Medical Devices”; Summarising some of the results of the 2012 Survey [6]](https://biomatdb.eu/wp-content/uploads/2022/12/BiomatDB-EUDAMED-A-database-to-strengthen-European-market-surveillance-and-increase-transparency-img-03.jpg)
Functionalities of EUDAMED
The purpose of the database is to provide the national competent authorities of EU Member States with access to relevant regulatory information and facilitate the implementation of a uniform approach to the Medical Device Directives. However, although authorities may record and have access to various market surveillance data, the web portal of EUDAMED is not fully functional yet.
Once finalized, the EUDAMED database will consist of six interconnected modules related to:
![Figure 3: Active and accessible to the public tabs at the EUDAMED website as of November 2022 [2]](https://biomatdb.eu/wp-content/uploads/2022/12/BiomatDB-EUDAMED-A-database-to-strengthen-European-market-surveillance-and-increase-transparency-img-04.jpg)
The use of EUDAMED is not yet mandatory for economic operators and notified bodies, and only the first three modules are already available. In contrast, the remaining modules (Vigilance, Clinical Investigation & Performance Studies and Market Surveillance) are under development and will be released once EUDAMED is announced as fully functional. The mandatory use of the system will start soon after a Commission notice is published in accordance with the transitional provisions set out in the medical devices regulations [11].
With consideration of the EU Medical Device Regulation (MDR), EUDAMED also addresses the emerging needs to:
- Enable identification and traceability of marketed medical devices;
- Store data about either active or completed clinical investigations;
- Collect trends of complaints and non-serious incidents related to medical devices;
- Collate serious incidents-related information;
- Actively monitor and meta-analyse trends in the information stored within the database;
- Update competent authorities on relevant developments.
![Figure 4: Article 33 – European database on medical devices, accessed November 2022 [3]](https://biomatdb.eu/wp-content/uploads/2022/12/BiomatDB-EUDAMED-A-database-to-strengthen-European-market-surveillance-and-increase-transparency-img-05.jpg)
Data protection and transparency
A Factsheet on MDR requirements for transparency and public information [10] is also available, addressing which aspects are shared in the EUDAMED public domain to ensure transparency for the patient, healthcare professionals, health authorities and industry.
In order to ensure data protection, the data controller of EUDAMED and its various electronic systems will be the Commission. Data protection is of utmost priority in Europe and the EUDAMED system abides by the GDPR’s rules. As stated in Article 33, “Eudamed shall contain personal data only insofar as necessary for the electronic systems referred to in paragraph 2 to collate and process information in accordance with this Regulation. Personal data shall be kept in a form which permits identification of data subjects for periods no longer than those referred to in Article 10(8)” [3]. Within their respective responsibilities, the Commission and the Member States shall ensure that inaccurate and unlawfully processed data are deleted per the applicable legislation.
Furthermore, the Global Medical Device Nomenclature is an essential tool for EUDAMED and a step towards harmonisation within the EU Member States. The GMDN is a collection of internationally agreed generic medical device descriptions used to define and identify medical devices. Thus, at the point of Notification within EUDAMED, each medical device has a GMDN code attached to it. This is mandated by the European Commission and has been the result of the collaborative effort of medical device experts from all over the world. It allows the identification of devices pertaining to performance and safety [12].
Finally, in practice, the introduction of EUDAMED will simplify notification procedures for manufacturers as they no longer need to register all in-vitro diagnostic devices in every member state before marketing them. Class IIa, Class IIb, and Class III medical devices only must be notified in Member States that require this registration in their transposition of the Medical Devices Directive.
References (English)
[1] European Commission (2010). Commission Decision of 10 April 2010 on the European Databank on Medical Devices (Eudamed). In: Official Journal of the European Union. [online] Available at: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2010:102:0045:0048:EN:PDF
[2] European Commission. EUDAMED Database. [online] Available at: https://ec.europa.eu/tools/eudamed/#/screen/home
[3] MDR: Article 33 – European database on medical devices. [online] Available at: https://www.medical-device-regulation.eu/2019/07/10/mdr-article-33-european-database-on-medical-devices/
[4] EUR-Lex (2017). Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017 on medical devices, amending Directive 2001/83/EC, Regulation (EC) No 178/2002 and Regulation (EC) No 1223/2009 and repealing Council Directives 90/385/EEC and 93/42/EEC. [online] Available at: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:32017R0745&locale=en
[5] EUR-Lex (2017). Regulation (EU) 2017/746 of the European Parliament and of the Council of 5 April 2017 on in vitro diagnostic medical devices and repealing Directive 98/79/EC and Commission Decision 2010/227/EU. [online] Available at: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:32017R0746&locale=en
[6] European Commission (2012). Evaluation of the “EUropean DAtabank on MEdical Devices”. [online] Available at: https://ec.europa.eu/docsroom/documents/12981/attachments/1/translations/en/renditions/pdf
[7] European Commission. Actor registration module. [online] Available at: https://health.ec.europa.eu/medical-devices-eudamed/actor-registration-module_en
[8] European Commission. UDI/Devices Registration. [online] Available at: https://health.ec.europa.eu/medical-devices-eudamed/udidevices-registration_en
[9] European Commission. Notified Bodies and Certificates module. [online] Available at: https://health.ec.europa.eu/medical-devices-eudamed/notified-bodies-and-certificates-module_en
[10] European Commission. Factsheet on MDR requirements for Transparency and Public Information. [online] Available at: https://health.ec.europa.eu/latest-updates/factsheet-mdr-requirements-transparency-and-public-information-2020-07-15_en
[11] European Commission. Overview. [online] Available at: https://health.ec.europa.eu/medical-devices-eudamed/overview_en
[12] Obelis Group (2019). What is Eudamed?. [online] Available at: https://www.obelis.net/news/what-is-eudamed/